Ocean Acidification Labs
Chemistry Bubble Protocol - Part Two
Roles:
Timer - Alexis Habib
Recorder - Faith Lobaton
Breather - Chris Muñoz
Materials - Megan Lambert
Central Question: How does Carbon Dioxide affect salt water?
Overview of Experiment
Exhaling carbon dioxide into a beaker of salt water mimics the gas exchange that occurs between Earth's atmosphere and oceans. You will track the resulting changes in ocean chemistry by monitoring changes in pH as you exhale into the salt water.
Introduction
Remember: C = Carbon, H = Hydrogen, O = Oxygen
When CO2 dissolves in seawater, the carbon and oxygen molecules bind together to form Carbonic Acid, which is shown in the following reaction:
CO2 + H2O → H2CO3
This Carbonic Acid breaks apart in the water, releasing hydrogen ions and bicarbonate, shown here:
H2CO3 → H+ + HCO3-
Student Pre-Lab Questions
1.) What gas are you blowing into the water?
CO2, our body absorbs Oxygen and releases Carbon Dioxide
2.) What happens to the gas when you blow it into the water?
It reacts with the water and gets absorbed by it, making the water react with the Carbon Dioxide and then releasing Hydrogen afterwards.
3.) How are you measuring change in the water during this lab?
We measure the change in the water with the universal indicator, that was previously stirred in with the solution, and changes color as the pH of the substance changes.
4.) What does measuring the pH of the water tell us?
The pH of the water tells us how much Hydrogen, or lack or Hydrogen at times, is in the water, the water becoming more acidic or basic depending on the pH value.
5.) After studying the reactions above, how do you think Carbonic Acid will affect the pH of salt water?
The Carbonic Acid added to the salt water will make the solution more acidic, lowering the pH of the total solution.
Hypothesis:
I believe that throughout the whole process, the pH of the solutions will continue to decline the whole time, becoming lighter with the universal indicator showing a visual aspect that show that the solution is becoming more acidic.
Student Post-Lab Questions
1.) As you blew through the straw, what were you adding to the water and how did that change the pH?
Carbon Dioxide, the Carbon Dioxide combining with the water and having that compound releasing Hydrogen into the atmosphere.
2.) What did the universal indicator tell us about the water?
3.) What does this tell us about the effects of Carbonic Acid in ocean water?
4.) Based on the results of your experimental protocol, which factor affects the pH of the water the most, temperature or salt?
The factor that affects the pH of the water the most would be
Conclusion:
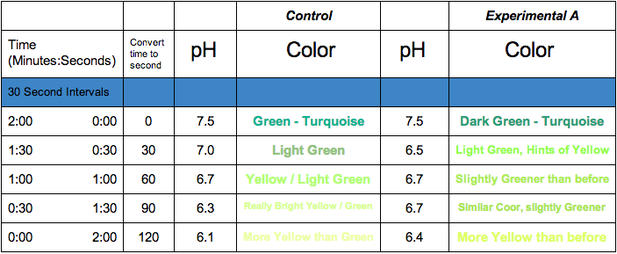
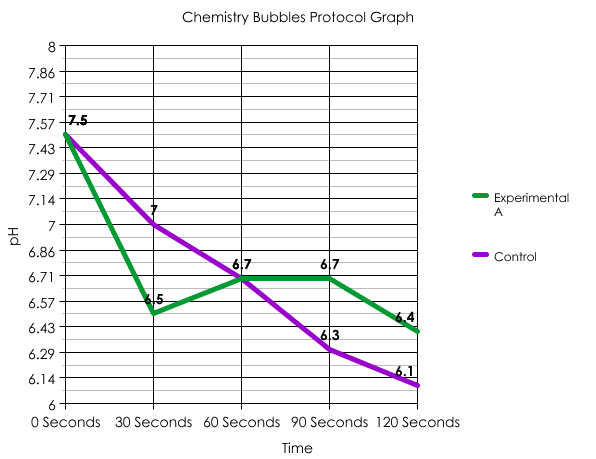
In conclusion, Experimental A was different from the Control because instead of steadily declining it slightly increased, as well as stayed constant in pH for a short period, stating my hypothesis false.
Roles:
Timer - Alexis Habib
Recorder - Faith Lobaton
Breather - Chris Muñoz
Materials - Megan Lambert
Central Question: How does Carbon Dioxide affect salt water?
Overview of Experiment
Exhaling carbon dioxide into a beaker of salt water mimics the gas exchange that occurs between Earth's atmosphere and oceans. You will track the resulting changes in ocean chemistry by monitoring changes in pH as you exhale into the salt water.
Introduction
Remember: C = Carbon, H = Hydrogen, O = Oxygen
When CO2 dissolves in seawater, the carbon and oxygen molecules bind together to form Carbonic Acid, which is shown in the following reaction:
CO2 + H2O → H2CO3
This Carbonic Acid breaks apart in the water, releasing hydrogen ions and bicarbonate, shown here:
H2CO3 → H+ + HCO3-
Student Pre-Lab Questions
1.) What gas are you blowing into the water?
CO2, our body absorbs Oxygen and releases Carbon Dioxide
2.) What happens to the gas when you blow it into the water?
It reacts with the water and gets absorbed by it, making the water react with the Carbon Dioxide and then releasing Hydrogen afterwards.
3.) How are you measuring change in the water during this lab?
We measure the change in the water with the universal indicator, that was previously stirred in with the solution, and changes color as the pH of the substance changes.
4.) What does measuring the pH of the water tell us?
The pH of the water tells us how much Hydrogen, or lack or Hydrogen at times, is in the water, the water becoming more acidic or basic depending on the pH value.
5.) After studying the reactions above, how do you think Carbonic Acid will affect the pH of salt water?
The Carbonic Acid added to the salt water will make the solution more acidic, lowering the pH of the total solution.
Hypothesis:
I believe that throughout the whole process, the pH of the solutions will continue to decline the whole time, becoming lighter with the universal indicator showing a visual aspect that show that the solution is becoming more acidic.
Student Post-Lab Questions
1.) As you blew through the straw, what were you adding to the water and how did that change the pH?
Carbon Dioxide, the Carbon Dioxide combining with the water and having that compound releasing Hydrogen into the atmosphere.
2.) What did the universal indicator tell us about the water?
3.) What does this tell us about the effects of Carbonic Acid in ocean water?
4.) Based on the results of your experimental protocol, which factor affects the pH of the water the most, temperature or salt?
The factor that affects the pH of the water the most would be
Conclusion:
In conclusion, Experimental A was different from the Control because instead of steadily declining it slightly increased, as well as stayed constant in pH for a short period, stating my hypothesis false.